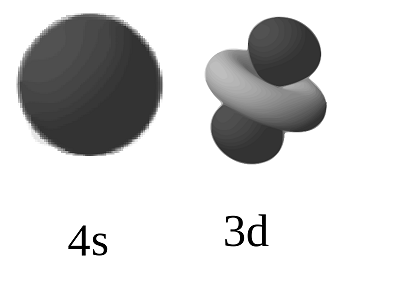
The fourth orbital looks the same but is orientated along the $x$- and $y$-axes. The first three orbitals are composed of 4 lobes each pointing along the coordinate system diagonals. There are five different d-orbitals due to the 5-fold degeneracy of m.
The orbitals are formed by a double lobe that point in the direction of the coordinate axes.įor $n \gt 2$ d-orbitals are possible. There are three different p-orbitals due to the 3-fold degeneracy of $m$. All s-orbitals are spherically symmetric.įor $n \gt 1$ the electron can be in an excited p-orbital as well. The 1s-orbital is lowest in energy and thus the ground state of the electron in a hydrogen atom. In some metals, where gap between bonding and anti-bonding orbitals is veruy nrrow they are formed in this gap. Usually these orbitals are results of symmetry conditions. This is the case in both the hydrogen atom and in the particle in a cube. Degenarate orbitals are orbitals having the same energies (eigenvalues). The different colors mark different signs of the wave function.įor each value of $n$ there is only one s-orbital since there is no $m$ degeneracy. If you have two or more coordinates that can be related by a symmetry operation then the system will contain degenerate states because swapping the coordinates associated with those degrees will result in a Hamiltonian with the same solutions. There are five d orbitals, four of which have a clover shape with different orientations, and one that is unique. There are three p orbitals that differ in orientation along a three-dimensional axis. In the notation (1s, 2p, 3d) the numbers represent the principal quantum number $n$ and the letters the angular momentum quantum numbers $l$. Each orbital type has a unique shape based on the energy of its electrons. This is done in the following for s-, p- and d-orbitals. One can visualise the orbital by creating three dimensional figures showing the areas with a high detection probability densitiy.
For historical reasons the different numbers are denoted by letters, namely $0 \rightarrow \text$ of the wave function at this particular location. The angular momentum quantum number $l = 0, 1, \dots, n-1$.The principal (energy) quantum number $n = 1,2,\dots$.As for a molecular system, I think it is best if i left. That is an example of degeneracy in an atomic system. Thus, the n2 energy is said to be degenerate since an electron can have this energy and be in a multitude of different eigenstates of the system. In quantum mechanics the state of a physical system is determined by its wave function $\psi$ which in turn is the solution of the Schrödinger equation for this system.įor a single electron in an atom such a particular state is called orbital and specified by three quantum numbers: Any electron in the s orbital state or one of the p-orbital states will have this energy. Quantum Mechanical Description Wave Function and Schrödinger Equation To that end a bit of quantum mechanics is required. This is however not possible without some knowledge about the atomic structure. Your reasoning regarding the additional l-term in V eff(r) giving the Y lm(Ω) functions a different energy is not correct b/c different l-values also affect the R nl(r) functions.The aim of this chapter is to understand the various bonds acting between atoms and molecules. I am pretty sure that we had this disucussion here a couple of times. Just google for hydrogen atom SO(4) and you will find numerous articles, scripts and presentations. The existence of the Laplace-Lenz-Runge vector and the l-degeneracy allowes one to solve the energy eigenvalue problem algebraically w/o solving the Schrödinger equation (W. The 1/r potential has not only the obvious SO(3) symmetry for spatial rotations but a larger SO(4) symmetry. the fact that E(n,l) = E(n) is l-independent is due to a hidden dynamical symmetry of the 1/r potential which results in an additional conserved quantity, the so-called Laplace-Lenz-Runge vector.
